
Second Academic Year for Medication Safety Scholars Program
March 22, 2024
Really excited to share the news that we will be beginning the second academic year for the Emily Jerry Foundation‘s Medication Safety Scholars Program! This comprehensive distance education and virtual engagement program was developed and successfully implemented over the… Read More



New York Scorecard
Grading Scale:
A – 85-100%, B – 70-84.9%, C – 55-69.9%, D – 40-54.9%, F – 0-39.9%
Grading Categories & Criteria
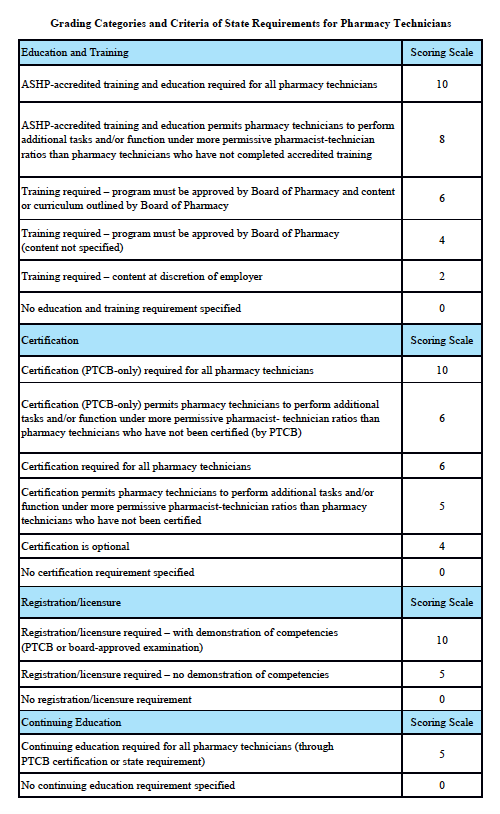
New York Law
I. LAWS
STATE OF NEW YORK
________________________________________________________________________
8319
2019-2020 Regular Sessions
IN ASSEMBLY
June 13, 2019
___________
Introduced by M. of A. ROMEO, GLICK, PEOPLES-STOKES, ENGLEBRIGHT,
MAGNARELLI, LIFTON, BENEDETTO, LUPARDO, STIRPE, BRONSON, FAHY, McDO-
NALD, SIMON, BICHOTTE, WALLACE, TAYLOR, BUTTENSCHON, EPSTEIN, McMAHON
— read once and referred to the Committee on Higher Education
AN ACT to amend the education law, in relation to the practice of regis-
tered pharmacy technician
The People of the State of New York, represented in Senate and Assem-
bly, do enact as follows:
1 Section 1. The education law is amended by adding a new article 137-A
2 to read as follows:
3 ARTICLE 137-A
4 REGISTERED PHARMACY TECHNICIANS
5 Section 6840. Introduction.
6 6841. Definition of the practice of registered pharmacy techni-
7 cian.
8 6842. Definitions.
9 6843. Practice of registered pharmacy technician and use of the
10 title “registered pharmacy technician”.
11 6844. Requirements for licensure as a registered pharmacy tech-
12 nician.
13 § 6840. Introduction. This article applies to the profession of regis-
14 tered pharmacy technician. The general provisions for all professions
15 contained in article one hundred thirty of this title shall apply to
16 this article.
17 § 6841. Definition of the practice of registered pharmacy technician.
18 1. A registered pharmacy technician may, under the direct personal
19 supervision of a licensed pharmacist, assist such licensed pharmacist,
20 as directed, in compounding, preparing, labeling, or dispensing of drugs
21 used to fill valid prescriptions or medication orders or in compounding,
22 preparing, and labeling in anticipation of a valid prescription or medi-
23 cation order for a patient to be served by the facility, in accordance
EXPLANATION–Matter in italics (underscored) is new; matter in brackets
[ ] is old law to be omitted.
LBD13315-02-9
A. 8319 2
1 with article one hundred thirty-seven of this title where such tasks
2 require no professional judgment. Such professional judgment shall only
3 be exercised by a licensed pharmacist. A registered pharmacy technician
4 may only practice in a facility licensed in accordance with article
5 twenty-eight of the public health law, or a pharmacy owned and operated
6 by such a facility, under the direct personal supervision of a licensed
7 pharmacist employed in such a facility or pharmacy. Such facility shall
8 be responsible for ensuring that the registered pharmacy technician has
9 received appropriate training to ensure competence before he or she
10 begins assisting a licensed pharmacist in compounding, preparing, label-
11 ing, or dispensing of drugs, in accordance with this article and article
12 one hundred thirty-seven of this title. For the purposes of this arti-
13 cle, direct personal supervision means supervision of procedures based
14 on instructions given directly by a supervising licensed pharmacist who
15 remains in the immediate area where the procedures are being performed,
16 authorizes the procedures and evaluates the procedures performed by the
17 registered pharmacy technicians and a supervising licensed pharmacist
18 shall approve all work performed by the registered pharmacy technician
19 prior to the actual dispensing of any drug.
20 2. In addition to the registered pharmacy technician services included
21 in subdivision one of this section, registered pharmacy technicians may
22 also assist a licensed pharmacist in the dispensing of drugs by perform-
23 ing the following functions that do not require a license under this
24 article:
25 a. receiving written or electronically transmitted prescriptions,
26 except that in the case of electronically transmitted prescriptions the
27 licensed pharmacist or pharmacy intern shall review the prescription to
28 determine whether in his or her professional judgment it shall be
29 accepted by the pharmacy, and if accepted, the licensed pharmacist or
30 pharmacy intern shall enter his or her initials into the records of the
31 pharmacy;
32 b. typing prescription labels;
33 c. keying prescription data for entry into a computer-generated file
34 or retrieving prescription data from the file, provided that such compu-
35 ter-generated file shall provide for verification of all information
36 needed to fill the prescription by a licensed pharmacist prior to the
37 dispensing of the prescription, meaning that the licensed pharmacist
38 shall review and approve such information and enter his or her initials
39 or other personal identifier into the recordkeeping system prior to the
40 dispensing of the prescription or of the prescription refill;
41 d. getting drugs from stock and returning them to stock;
42 e. getting prescription files and other manual records from storage
43 and locating prescriptions;
44 f. counting dosage units of drugs;
45 g. placing dosage units of drugs in appropriate containers;
46 h. affixing the prescription label to the containers;
47 i. preparing manual records of dispensing for the signature or
48 initials of the licensed pharmacist;
49 j. handing or delivering completed prescriptions to the patient or the
50 person authorized to act on behalf of the patient and, in accordance
51 with the relevant commissioner’s regulations, advising the patient or
52 person authorized to act on behalf of the patient of the availability of
53 counseling to be conducted by the licensed pharmacist or pharmacy
54 intern; or
55 k. performing other functions as defined by the commissioner’s regu-
56 lations.
A. 8319 3
1 3. Under the direct personal supervision of a licensed pharmacist,
2 unlicensed persons who are not registered pharmacy technicians may
3 assist licensed pharmacists in performing tasks that do not require
4 licensure in accordance with regulations promulgated by the commissioner
5 and are also described in subdivision two of this section. Unlicensed
6 persons who are not registered pharmacy technicians shall not engage in
7 or assist in compounding.
8 4. No licensed pharmacist shall obtain the assistance of more than two
9 registered pharmacy technicians in the performance of licensed tasks
10 within their scope of practice or four unlicensed persons, in the
11 performance of the activities that do not require licensure, the total
12 of such persons shall not exceed four individuals at any one time.
13 Pharmacy interns shall be exempt from such ratios, but shall be super-
14 vised in accordance with commissioner’s regulations. Individuals who are
15 responsible for the act of placing drugs which are in unit-dose packag-
16 ing into medication carts as part of an approved unit-dose drug distrib-
17 ution system for patients in institutional settings shall be exempt from
18 such ratio, provided that such individuals are not also engaged in
19 performing the activities set forth in subdivision one or paragraph b,
20 c, d, e, f, g, h, or i of subdivision two of this section. The licensed
21 pharmacist shall provide the degree of supervision of such persons as
22 may be appropriate to ensure compliance with the relevant provisions of
23 regulations of the commissioner.
24 § 6842. Definitions. As used in this article:
25 1. “Licensed pharmacist” means a person licensed to practice pharmacy
26 pursuant to article one hundred thirty-seven of this title.
27 2. “Pharmacy intern” means a person practicing under a limited permit
28 pursuant to section sixty-eight hundred six of this title.
29 3. “Professional judgment” means professional decision-making by a
30 licensed pharmacist, including, but not limited to, such activities as:
31 a. interpreting a prescription or medication order for therapeutic
32 acceptability and appropriateness or engaging in the calculations behind
33 any such formulations;
34 b. interpreting and evaluating a prescription or medication order for
35 conformance with legal requirements, authenticity, accuracy and inter-
36 action of the prescribed drug with other known prescribed and over-the
37 -counter drugs;
38 c. receiving oral prescriptions from prescribers; or
39 d. counseling patients.
40 4. “Compounding” means the combining, admixing, mixing, diluting,
41 pooling, reconstituting, or otherwise altering of a drug or bulk drug
42 substance to create a drug.
43 5. “Drugs”, “pharmacopeia”, “labeling” and “sterile drug” shall have
44 the same definitions as set forth in section sixty-eight hundred two of
45 this title.
46 § 6843. Practice of registered pharmacy technician and use of the
47 title “registered pharmacy technician”. Only a person licensed to prac-
48 tice as a registered pharmacy technician under this article or otherwise
49 authorized shall practice as a registered pharmacy technician or use the
50 title “registered pharmacy technician.”
51 § 6844. Requirements for licensure as a registered pharmacy techni-
52 cian. To qualify for licensure as a “registered pharmacy technician”, an
53 applicant shall fulfill the following requirements:
54 1. Application: file an application with the department;
55 2. Education: have received an education, including high school gradu-
56 ation or its equivalent, as determined by the department;
A. 8319 4
1 3. Certification from a nationally accredited pharmacy technician
2 certification program acceptable to the department;
3 4. Age: at the time of application be at least eighteen years of age;
4 5. Character: be of good moral character as determined by the depart-
5 ment; and
6 6. Fee: pay a fee determined by the department for initial license and
7 for each triennial registration period.
8 § 2. The education law is amended by adding a new section 6832 to read
9 as follows:
10 § 6832. Limitations on assistance of an unlicensed person. 1. Subject
11 to the limitations set forth in subdivision two of this section, an
12 unlicensed person may assist a licensed pharmacist in the dispensing of
13 drugs by:
14 (a) receiving written or electronically transmitted prescriptions,
15 except that in the case of electronically transmitted prescriptions the
16 licensed pharmacist or pharmacy intern shall review the prescription to
17 determine whether in his or her professional judgment it shall be
18 accepted by the pharmacy, and if accepted, the licensed pharmacist or
19 pharmacy intern shall enter his or her initials into the records of the
20 pharmacy;
21 (b) typing prescription labels;
22 (c) keying prescription data for entry into a computer-generated file
23 or retrieving prescription data from the file, provided that such compu-
24 ter-generated file shall provide for verification of all information
25 needed to fill the prescription by a licensed pharmacist prior to the
26 dispensing of the prescription, meaning that the licensed pharmacist
27 shall review and approve such information and enter his or her initials
28 or other personal identifier into the recordkeeping system prior to the
29 dispensing of the prescription or of the prescription refill;
30 (d) getting drugs from stock and returning them to stock;
31 (e) getting prescription files and other manual records from storage
32 and locating prescriptions;
33 (f) counting dosage units of drugs;
34 (g) placing dosage units of drugs in appropriate containers;
35 (h) affixing the prescription label to the containers;
36 (i) preparing manual records of dispensing for the signature or
37 initials of the licensed pharmacist;
38 (j) handing or delivering completed prescriptions to the patient or
39 the person authorized to act on behalf of the patient and, in accordance
40 with the relevant commissioner’s regulations, advising the patient or
41 person authorized to act on behalf of the patient of the availability of
42 counseling to be conducted by the licensed pharmacist or pharmacy
43 intern; and
44 (k) performing other functions as defined by the commissioner’s regu-
45 lations.
46 2. Except for a licensed pharmacist employed by a facility licensed in
47 accordance with article twenty-eight of the public health law or a phar-
48 macy owned and operated by such a facility, as defined in article one
49 hundred thirty-seven-A of this title, no licensed pharmacist shall
50 obtain the assistance of more than four unlicensed persons, in the
51 performance of the activities that do not require licensure, the total
52 of such persons shall not exceed four individuals at any one time. Phar-
53 macy interns shall be exempt from such ratios, but shall be supervised
54 in accordance with the commissioner’s regulations. Individuals who are
55 responsible for the act of placing drugs which are in unit-dose packag-
56 ing into medication carts as part of an approved unit-dose drug distrib-
A. 8319 5
1 ution system for patients in institutional settings shall be exempt from
2 such ratio, provided that such individuals are not also engaged in
3 performing the activities set forth in paragraph (b), (c), (d), (e),
4 (f), (g), (h) or (i) of subdivision one of this section. The licensed
5 pharmacist shall provide the degree of supervision of such persons as
6 may be appropriate to ensure compliance with the relevant provisions of
7 regulations of the commissioner.
8 § 3. Section 6804 of the education law, as added by chapter 987 of the
9 laws of 1971, is amended to read as follows:
10 § 6804. State board of pharmacy. A state board of pharmacy shall be
11 appointed by the regents on recommendation of the commissioner for the
12 purpose of assisting the regents and the department on matters of
13 professional licensing and professional conduct in accordance with
14 section sixty-five hundred eight of this title. The board shall be
15 composed of not less than nine pharmacists licensed in this state for at
16 least five years and two registered pharmacy technicians. The initial
17 registered pharmacy technician members of the state board of pharmacy
18 need not be licensed prior to their appointment but shall have met all
19 other requirements of licensure pursuant to section sixty-eight hundred
20 forty-two of this title except for filing an application and paying a
21 fee. An executive secretary to the board shall be appointed by the
22 regents on recommendation of the commissioner and shall be a pharmacist
23 licensed in this state for at least five years. The board shall [have
24 power] provide assistance to the department:
25 a. To regulate the practice of pharmacy, registered pharmacy techni-
26 cians and the employment of interns and employees in pharmacies,
27 b. To regulate and control the sale, distribution, character and stan-
28 dard of drugs, poisons, cosmetics, devices and new drugs,
29 c. [To employ inspectors and chemists,
30 d.] To prevent the sale or distribution of such drugs, poisons,
31 cosmetics, devices and new drugs as do not conform to the provisions of
32 this article or of the public health law,
33 [e.] d. To investigate alleged violations of the provisions of this
34 article[, through its own investigative personnel or those of other
35 agencies, to conduct hearings, to levy money penalties, and to bring
36 alleged violations to the notice of the attorney general], and
37 [f.] e. To issue limited permits or registrations.
38 § 4. This act shall take effect eighteen months after it shall have
39 become a law; provided, however, that the department of education is
40 authorized to promulgate regulations that may be required and to take
41 other measures necessary to implement the provisions of this act prior
42 to such effective date, including appointment of two registered pharmacy
43 technicians to the state board of pharmacy, and the acceptance and proc-
44 essing of applications for licensure.
The data contained in this Annual Scorecard are accurate as of April 2020 . Because statutes and regulations are continually revised, the data are subject to change. These data have been verified with the state board of pharmacy. This scorecard is updated on an annual basis in order to incorporate statutory and regulatory changes. A new scorecard will be issued in each year if updates are made.
Scoring rationale for Education and Training:
In order to protect the public and help ensure patient safety, it is important that pharmacy technicians are properly educated and trained. The most rigorous training is accredited training. The sole entity empowered to accredit pharmacy-technician training programs is the American Society of Health-System Pharmacists (ASHP). Please note that this is “programmatic accreditation” – not “institutional accreditation.” It is the content of the training program – as measured against established standards – that is being evaluated and accredited. Accredited training is vital to protecting patient safety because it means that a pharmacy-technician training program has met established quality standards to provide assurance and confidence to the public. For more information, please see http://www.ashp.org/menu/Accreditation/TechnicianAccreditation.aspx.
Scoring rationale for Certification:
Certification is the process by which a nongovernmental agency or association grants recognition to an individual who has met certain predetermined qualifications specified by that agency or association. This is often determined by an examination process. Numerous organizations have recommended that the certification exam conducted by the Pharmacy Technician Certification Board (PTCB) should be recognized as the sole, nationally-accredited certification exam for pharmacy technician certification – including the National Association of Boards of Pharmacy (NABP), the Texas State Board of Pharmacy (TSBP), and the U.S. Department of Veterans Affairs (VA). In a recent report, NABP recommended that states be encouraged to “recognize certification by the Pharmacy Technician Certification Board (PTCB).” Moreover, NABP performed a psychometric audit of the PTCB’s pharmacy technician certification examination (PTCE) in 2001 and determined that the PTCE is psychometrically sound, defensible, and valid. In May 2010, the TSBP awarded the PTCB with the Pharmacy Technician Certification Provider contract in Texas. PTCB was selected for the contract after a rigorous bidding and evaluation process that included formal reviews and evaluations from three independent psychometricians. TSBP confidently recognizes PTCB as the single provider of certification examinations for pharmacy technicians. In addition, in June 2010, the VA began requiring PTCB certification for VA pharmacy technicians employed at grade GS-6 and above.
Scoring rationale for Registration/Licensure:
Registration/licensure is the process by which the state maintains a list of all pharmacy technicians in the state and grants permission for an individual to work as a pharmacy technician in the state based on the applicant’s completion of all pre-requisites to registration/licensure – such as required training and certification.
Scoring rationale for Continuing Education:
Continuing education enables pharmacy technicians to fulfill their professional responsibility to maintain competence and up-to-date knowledge and skills in an environment of technological advances and increasingly complex, new medications and therapies.
Our Mission
The Emily Jerry Foundation is determined to help make our nation’s, world renowned, medical facilities safer for everyone, beginning with our babies and children. We are accomplishing this very important objective by focusing on increasing public awareness of key patient safety related issues and identifying technology and best practices that are proven to minimize the “human error” component of medicine. Through our ongoing efforts The Emily Jerry Foundation is working hard to save lives every day.
Recent Posts
Archives