
EJF Keynote at the South Carolina Society of Health-System Pharmacists (SCSHP) 2026 Annual Meeting
March 27, 2026
By ejfadmin
Twenty years ago, I lost my daughter Emily to a preventable medication error. The week before last, I stood on Kiawah Island delivering the keynote at the South Carolina Society of Health-System Pharmacists (SCSHP) 2026 Annual Meeting…surrounded by… Read More



Wisconsin Scorecard
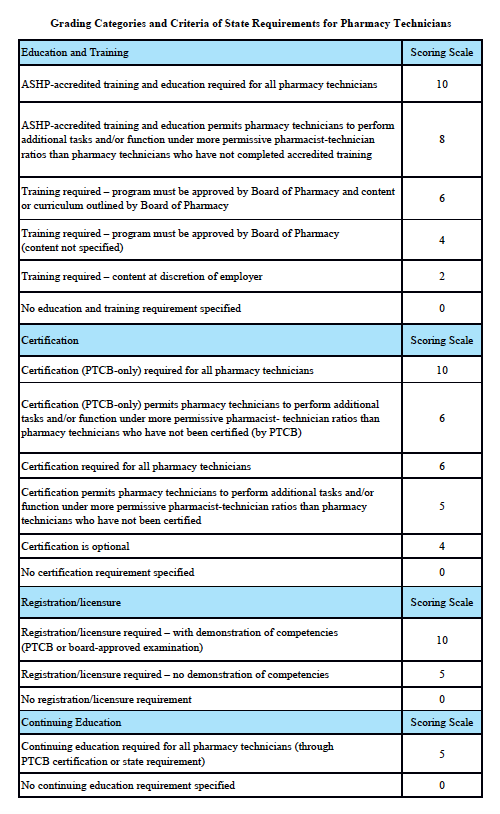
Grading Scale:
A – 85-100%, B – 70-84.9%, C – 55-69.9%, D – 40-54.9%, F – 0-39.9%
Grading Categories & Criteria
Wisconsin Law
I. Laws
Wisconsin Statutes
https://docs.legis.wisconsin.gov/statutes/statutes/450
450.068 Pharmacy technicians; registration.
(1) No person may engage in the practice of a pharmacy technician or use the title “pharmacy technician” or “pharmacy tech” unless the person is registered as a pharmacy technician by the board.
(2) Except as provided in s. 450.10, the board shall issue a registration as a pharmacy technician to an applicant who satisfies all of the following:
(a) The applicant submits an application for registration on a form provided by the board that specifies all of the following:
1. The applicant’s home address.
2. If the applicant is employed, the name and address of the applicant’s employer, and the applicant’s place of employment.
(b) The applicant satisfies one of the following:
1. The applicant is at least 18 years of age and has graduated from high school or has attained high school graduation equivalency as determined by the department of public instruction.
2. The applicant is enrolled in a youth apprenticeship program for pharmacy technicians that is on the list of youth apprenticeship programs approved by the department of workforce development under s. 106.13 (2m).
(d) The applicant pays the fee specified in s. 440.05 (1).
(3) A pharmacy technician shall do all of the following:
(a) Notwithstanding s. 440.11, if the pharmacy technician moves from the last address provided to the department, notify the department of his or her new address within 10 days of the change in writing or in accordance with other notification procedures approved by the department.
(b) If the pharmacy technician changes his or her employer or place of employment, notify the department of his or her new employer or address of employment within 10 days of the change in writing or in accordance with other notification procedures approved by the department and, if required by the department, pay the transfer fee under s. 440.05 (7).
History: 2021 a. 100.
Wisconsin Administrative Code
https://docs.legis.wisconsin.gov/code/admin_code/phar/7
Phar 1.02 Definitions
(11m) “Pharmacy technician” means a person registered by the board under s. 450.068, Stats.
Phar 5.07 Pharmacy technicians.
(1) All requirements for renewal and reinstatement of a pharmacy technician registration are specified in ch. Phar 19.
(2) No pharmacist whose license has been denied, revoked, suspended, or restricted for disciplinary purposes is eligible to be registered as a pharmacy technician.
History: EmR2303: emerg. cr., eff. 2-3-23; CR 23-072: cr. Register August 2024 No. 824, eff. 9-1-24.
Chapter Phar 19 REGISTRATION OF PHARMACY TECHNICIANS
Phar 19.01 Registration.
(1) No person may engage in the practice of a pharmacy technician or use the title “pharmacy technician” or “pharmacy tech” unless the person is registered as a pharmacy technician by the board.
(2) A person applying for a pharmacy technician registration shall satisfy all of the following:
(a) Submit a completed application form.
Note: Instructions for applications are available on the department of safety and professional services’ website at http://dsps.wi.gov.
(b) Pay the fee determined by the department under s. 440.05 (1), Stats.
(c) Subject to ss. 111.321, 111.322, and 111.335, Stats., the applicant does not have an arrest or conviction record.
(d) The applicant satisfies one of the following:
1. Is at least 18 years of age and has graduated from high school or has attained high school graduation equivalency as determined by the department of public instruction.
2. Is enrolled in a youth apprenticeship program for pharmacy technicians that is on the list of youth apprenticeship programs approved by the department of workforce development under s. 106.13 (2m), Stats.
(3) A person who has applied for a registration as a pharmacy technician and whose practice as a pharmacy technician is limited to performing duties under the direct supervision of a person licensed as a pharmacist by the board and during the period before which the board takes final action on the persons application may practice as a pharmacy technician.
History: EmR2303: emerg. cr., eff. 2-3-23; CR 23-072: cr. Register August 2024 No. 824, eff. 9-1-24.
Phar 19.02 Scope of practice. A pharmacy technician may administer vaccines as authorized under s. 450.035 (2h), Stats., and may perform technical dispensing functions, compounding, packaging, labeling and storage, pharmacy and inventory management, and other activities involved in the practice of pharmacy delegated by a pharmacist. A pharmacy technician may not perform any of the following:
(1) Except as allowed under s. Phar 7.14, provide the final verification for the accuracy, validity, completeness, or appropriateness of the filled prescription or medication order.
(2) Complete the drug utilization review under s. Phar 7.03.
(3) Administer any prescribed drug products, or devices under s. 450.035 (1t), Stats.
(4) Provide patient specific counseling or consultation.
(5) Make therapeutic alternate drug selections.
Phar 19.02(6)(6) Provide supervision over the practice of pharmacy to other pharmacy technicians.
History: EmR2303: emerg. cr., eff. 2-3-23; CR 23-072: cr. Register August 2024 No. 824, eff. 9-1-24; correction in (intro.) made under s. 35.17, Stats., Register August 2024 No. 824.
Phar 7.14 Pharmacy product verification technician-check-pharmacy technician.
(1) Definitions. In this section:
(a) “Pharmacy product verification technician” means a registered pharmacy technician to whom the pharmacist has delegated the task of product verification.
(b) “Pharmacy product verification technician-check- pharmacy technician” means the process in which a pharmacy product verification technician conducts the task of product verification of technical dispensing functions completed by a pharmacy technician. A pharmacy product verification technician may not conduct product verification as part of the final check of their own product preparation.
(c) “Product verification” means doing a check of the accuracy and correctness of a product, including drug, strength, formulation, as part of the final check and ensure the product has not reached its expiration or beyond use date.
(d) “Supervising pharmacist” means the pharmacist licensed in this state, who is responsible for the operations and outcomes of product verification done by a pharmacy product verification technician and ensuring for direct supervision of the pharmacy product verification technician.
(2) Pharmacy product verification technician qualifications. A pharmacist may delegate the product verification of a prescription or chart order to a pharmacy technician who meets all of the following:
(b) Completed an accredited pharmacy technician training program or has a minimum of 500 hours of experience in product selection, labeling and packaging.
(c) Completed a didactic and practical training curriculum approved by the supervising and managing pharmacist that includes training in all of the following:
1. Elements of correct product including all of the following:
a. Drug name.
b. Strength.
c. Formulation.
d. Expiration date.
e. Beyond use date.
2. Common dispensing medication errors and concepts including all of the following:
a. Wrong medication.
b. Wrong strength.
c. Wrong formulation.
d. Extra or insufficient quantity.
e. Omitted medications if utilizing unit dose or compliance packaging.
f. Expired medication.
g. Look-alike or sound-alike errors.
h. High-alert medications.
3. Eligible products for pharmacy product verification technician-check-pharmacy technician.
4. Organizational policies and procedures on reporting of medication errors.
5. Overview of the medication use process including all of the following:
a. Procurement.
b. Ordering.
c. Dispensing.
d. Administration.
e. Monitoring.
6. A practical training designed to assess the competency of the pharmacy technician prior to starting the validation process. The practical training shall include simulation of at least 2 occurrences of each of the following:
a. Wrong drug.
b. Wrong strength.
c. Wrong formulation.
d. Omitted medication, if utilizing unit dose or compliance packaging.
(d) Completed the following validation process:
1. The pharmacy technician being validated shall make a product verification on the work of a pharmacist or another pharmacy technician for accuracy and correctness of a minimum of 500 product verifications over a minimum of 5 separate days and achieve an accuracy rate of at least 99.8%.
2. A pharmacist shall audit 100% of the product verifications made by the pharmacy technician during the validation process.
(e) Notwithstanding pars. (b) to (d), an individual who completed the board’s pilot program validation process between October 1, 2016 and September 30, 2019, meets the pharmacy product verification technician qualifications unless the individual fails to meet the quality assurance standards under sub. (4).
(3) Eligible product.
(a) Institutional pharmacies. The pharmacy product verification technician may do the product verification in an institutional pharmacy if all of the following requirements are met:
1. The source drug product or device is in an original package from a manufacturer or a licensed pharmacist has ensured that the source package is labeled with the correct name, strength, form, control or lot number, and beyond use or expiration date.
2. A drug utilization review performed by a pharmacist prior to dispensing.
3. The drug product will be administered by an individual authorized to administer medications at the institution where the medication is administered.
(b) Community pharmacies. The pharmacy product verification technician may do the product verification in a community pharmacy if all of the following requirements are met:
1. The source drug product or device is in an original package from a manufacturer or a licensed pharmacist has ensured that the source package is labeled with the correct name, strength, form, control or lot number, and beyond use or expiration date.
2. A drug utilization review performed by a pharmacist prior to dispensing.
3. A non-pharmacist shall be able to check the accuracy of the medication by one of the following:
a. The drug product or device is in the original packaging from a manufacturer.
b. The drug product or device includes a description of the drug product or device on the prescription label.
c. The pharmacist shows the patient or patient’s agent the drug product or device and provides a monograph that includes a description of the drug product or device.
(4) Quality assurance.
(a) A minimum of 5% of each pharmacy product verification technician’s verifications shall be audited by a licensed pharmacist. The accuracy of each pharmacy product verification technician shall be tracked individually.
(b) A record of each pharmacy product verification technician-check-pharmacy technician audit shall include all of the following:
1. Name of the pharmacy product verification technician.
2. Total number of product verifications performed.
3. Number of product verifications audited by the pharmacist.
4. Percentage of product verifications audited by pharmacist.
5. Percentage of accuracy.
6. Number of product verification errors identified.
7. Type of error under sub. (2) (c) 2. a. to c. and e.
(c) On a quarterly basis, the supervising pharmacist shall perform an assessment of each pharmacy product verification technician’s previous 12 months accuracy and correctness of pharmacy product verifications including a review of the quality assurance log.
(d) A pharmacy product verification technician shall be revalidated if the individual fails to maintain a product verification accuracy rate of 99.8% based on the quarterly assessment of the previous 12 months or has not performed verifications within the last 6 months.
(5) Policies and procedures. Each pharmacy shall maintain policies, procedures, and training materials for the pharmacy product verification by technicians which shall be made available to the board upon request.
(6) Records.
(a) Each pharmacy shall maintain for 5 years the following records:
1. All validation records of each pharmacy product verification technician that include the dates that the validation occurred, the number of product verifications performed, the number of product verification errors, and overall accuracy rate.
2. Documentation indicating accepting responsibility for compliance with this section, signed and dated by both the managing pharmacist and supervising pharmacist, indicating the name of the supervising pharmacist, and the dates the supervision responsibilities begin and end.
3. Quality assurance audits and quarterly assessments.
(b) Records shall be made available to the board upon request.
History: CR 19-145: cr. Register December 2020 No. 780, eff. 1-1-21; corrections in (2) (e) made under ss. 13.92 (4) (b) 12. and 35.17, Stats., and correction in (2) (c) 6. (intro.), (4) (b) 7. made under s. 35.17, Stats., Register December 2020 No. 780; EmR2303: emerg. am. (title), (1) (a), (b), (d), (2) (intro.), r. (2) (a), am. (2) (b), (c) 3., 6., (d) 1., 2., (e), (3) (a) (intro.), (b), (4) (a), (b) (intro.), 1., (c), (d), (5), (6) (a) 1., 2., eff. 2-3-23; CR 23-072: r. and recr. (title), am. (1) (a), (b), (d), (2) (intro.), r. (2) (a), am. (2) (b), (c) 3., 6., (d) 1., 2., (e), (3) (a) (intro.), (b) (intro.), 1., (c), (d), (5), (6) (a) 1., 2. Register August 2024 No. 824, eff. 9-1-24.
”
The data contained in this 2012 Annual Scorecard are accurate as of December 2012 . Because statutes and regulations are continually revised, the data are subject to change. These data have been verified with the state board of pharmacy. This scorecard is updated on an annual basis in order to incorporate statutory and regulatory changes. A new scorecard will be issued in July 2013.
Scoring rationale for Education and Training:
In order to protect the public and help ensure patient safety, it is important that pharmacy technicians are properly educated and trained. The most rigorous training is accredited training. The sole entity empowered to accredit pharmacy-technician training programs is the American Society of Health-System Pharmacists (ASHP). Please note that this is “programmatic accreditation” – not “institutional accreditation.” It is the content of the training program – as measured against established standards – that is being evaluated and accredited. Accredited training is vital to protecting patient safety because it means that a pharmacy-technician training program has met established quality standards to provide assurance and confidence to the public. For more information, please see http://www.ashp.org/menu/Accreditation/TechnicianAccreditation.aspx.
Scoring rationale for Certification:
Certification is the process by which a nongovernmental agency or association grants recognition to an individual who has met certain predetermined qualifications specified by that agency or association. This is often determined by an examination process. Numerous organizations have recommended that the certification exam conducted by the Pharmacy Technician Certification Board (PTCB) should be recognized as the sole, nationally-accredited certification exam for pharmacy technician certification – including the National Association of Boards of Pharmacy (NABP), the Texas State Board of Pharmacy (TSBP), and the U.S. Department of Veterans Affairs (VA). In a recent report, NABP recommended that states be encouraged to “recognize certification by the Pharmacy Technician Certification Board (PTCB).” Moreover, NABP performed a psychometric audit of the PTCB’s pharmacy technician certification examination (PTCE) in 2001 and determined that the PTCE is psychometrically sound, defensible, and valid. In May 2010, the TSBP awarded the PTCB with the Pharmacy Technician Certification Provider contract in Texas. PTCB was selected for the contract after a rigorous bidding and evaluation process that included formal reviews and evaluations from three independent psychometricians. TSBP confidently recognizes PTCB as the single provider of certification examinations for pharmacy technicians. In addition, in June 2010, the VA began requiring PTCB certification for VA pharmacy technicians employed at grade GS-6 and above.
Scoring rationale for Registration/Licensure:
Registration/licensure is the process by which the state maintains a list of all pharmacy technicians in the state and grants permission for an individual to work as a pharmacy technician in the state based on the applicant’s completion of all pre-requisites to registration/licensure – such as required training and certification.
Scoring rationale for Continuing Education:
Continuing education enables pharmacy technicians to fulfill their professional responsibility to maintain competence and up-to-date knowledge and skills in an environment of technological advances and increasingly complex, new medications and therapies.
Our Mission
The Emily Jerry Foundation is determined to help make our nation’s, world renowned, medical facilities safer for everyone, beginning with our babies and children. We are accomplishing this very important objective by focusing on increasing public awareness of key patient safety related issues and identifying technology and best practices that are proven to minimize the “human error” component of medicine. Through our ongoing efforts The Emily Jerry Foundation is working hard to save lives every day.
Recent Posts
Archives