
EJF Keynote at the South Carolina Society of Health-System Pharmacists (SCSHP) 2026 Annual Meeting
March 27, 2026
By ejfadmin
Twenty years ago, I lost my daughter Emily to a preventable medication error. The week before last, I stood on Kiawah Island delivering the keynote at the South Carolina Society of Health-System Pharmacists (SCSHP) 2026 Annual Meeting…surrounded by… Read More



Alaska Scorecard
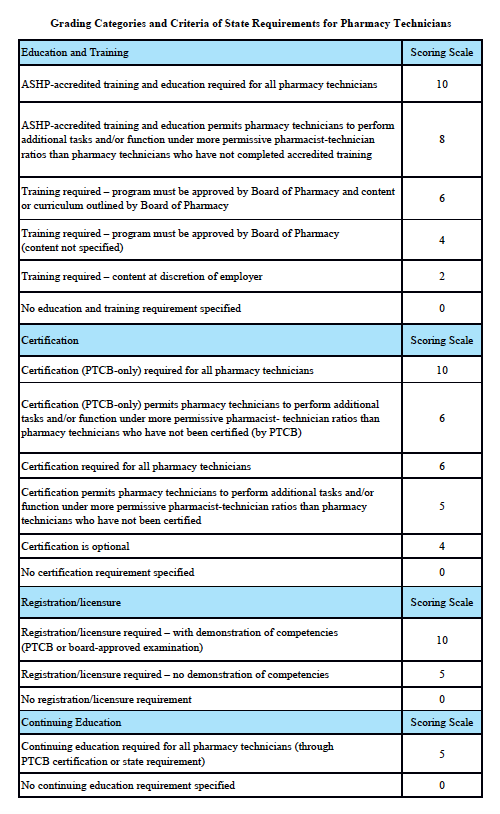
Grading Scale:
A – 85-100%, B – 70-84.9%, C – 55-69.9%, D – 40-54.9%, F – 0-39.9%
Grading Categories & Criteria
Alaska Law
I. Laws
Board of Pharmacy Statutes (08.80) and Regulations (12 AAC 52)
https://www.commerce.alaska.gov/web/Portals/5/pub/PharmacyStatutes.pdf
Sec. 08.80.030. Powers and duties of the board.
If only highlighting paragraph (b)(9), then recommend deleting all preceding clauses. If intending to include the entire section, recommend adding the remaining clauses (b)(12)-(b)(18).
(a) The board is responsible for the control and regulation of the practice of pharmacy.
(b) In order to fulfill its responsibilities, the board has the powers necessary for implementation and enforcement of this chapter, including the power to
(9) license and regulate the training, qualifications, and employment of pharmacy interns and pharmacy
technicians;
Sec. 08.80.160. FEES.
No change.
Sec. 08.80.160. Fees. The Department of Commerce, Community, and Economic Development shall set fees
under AS 08.01.065 for the following:
(1) examination;
(2) reexamination;
(3) investigation for licensing by license transfer;
(4) pharmacist license;
(5) temporary license;
(6) pharmacy technician license;
(7) pharmacy intern license;
(8) emergency permit;
(9) license amendment or replacement;
(10) licensure of a facility classified under AS 08.80.157(b).
Sec. 08.80.165. Continuing education requirements. The board shall establish requirements for continuing
education in pharmacy that must be satisfied before a license issued under this chapter may be renewed.
” “There are several more definitions now and one deleted (prospective drug use review). Recommend truncating this section to only those that apply to pharmacy technicians, such as (26).
Sec. 08.80.480. DEFINITIONS.
In this chapter, unless the context otherwise requires,
(1) “administer” means the direct application of a drug to the body of a patient or research subject by
injection, inhalation, ingestion, or other means;
(2) “biological product” means a product that is applicable to the prevention, treatment, or cure of a disease or
condition of human beings, and is a virus, therapeutic serum, toxin, antitoxin, vaccine, blood, blood component or
derivative, allergenic product, protein other than a chemically synthesized polypeptide, or analogous product, or
arsphenamine or derivative of arsphenamine or any other trivalent organic arsenic compound;
(3) “board” means the Board of Pharmacy;
(4) “compounding” means the preparation, mixing, assembling, packaging, or labeling of a drug or device (A)
as the result of a practitioner’s prescription drug order or initiative based on the relationship of the practitioner,
patient, and pharmacist in the course of professional practice or (B) for the purpose of, or as an incident to, research,
teaching, or chemical analysis and not for sale or dispensing; “compounding” also includes the preparation of drugs
or devices in anticipation of prescription drug orders based on routine, regularly observed prescribing patterns;
(5) “controlled substance” has the meaning given in AS 11.71.900;
(6) “deliver” or “delivery” means the actual, constructive, or attempted transfer of a drug or device from one
person to another, whether or not for consideration;
(7) “device” means an instrument, apparatus, implement, machine, contrivance, implant, or other similar or
related article, including a component part or accessory, that is required under federal law to bear the label “Caution:
Federal or state law requires dispensing by or on the order of a physician”;
(8) “dispense” or “dispensing” means the preparation and delivery of a drug or device to a patient or patient’s
agent under a lawful order of a practitioner in a suitable container appropriately labeled for subsequent
administration to, or use by, a patient;
(9) “distribute” means the delivery of a drug or device other than by administering or dispensing;
(10) “drug” means an article recognized as a drug in an official compendium, or supplement to an official
compendium; an article intended for use in the diagnosis, cure, mitigation, treatment, or prevention of disease in
man or animal; an article other than food, intended to affect the structure or function of the body of man or animal;
and an article intended for use as a component of an article specified in this paragraph but does not include devices
or their components, parts, or accessories;
(11) “drug regimen review” includes evaluation of the prescription drug order and patient record for
(A) known allergies;
(B) rational therapy-contraindications;
(C) reasonable dose and route of administration;
(D) reasonable directions for use;
(E) duplication of therapy;
(F) drug-drug, drug-food, and drug-disease interactions;
(G) adverse drug reactions; and
(H) proper utilization, including over- or under-utilization, and optimum therapeutic outcomes;
(12) “equivalent drug product” means a drug product that has the same established name, active ingredients,
strength or concentration, and route of administration and that is formulated to contain the same amount of active
ingredients in the same dosage form and to meet the same compendia or other applicable standards for strength,
quality, purity, and identity, but that may differ in characteristics such as shape, scoring configuration, packaging,
excipients including colors, flavors, preservatives, and expiration time;
(13) “interchangeable biological product” means a biological product that the United States Food and Drug
Administration has determined
(A) meets the standards for interchangeability under 42 U.S.C. 262(k)(4); or
(B) is therapeutically equivalent to another biological product under the most recent edition or supplement
of the United States Food and Drug Administration’s Approved Drug Products with Therapeutic Equivalence
Evaluations;
(14) “intern” means an individual who is
(A) currently licensed by this state to engage in the practice of pharmacy while under the personal
supervision of a pharmacist and is satisfactorily progressing toward meeting the requirements for licensure as a
pharmacist; or
(B) a graduate from a college of pharmacy who is currently licensed by the board for the purpose of
obtaining practical experience as a requirement for licensure as a pharmacist;
(15) “labeling” means the process of preparing and affixing a label to a drug container, exclusive, however, of
the labeling by a manufacturer, packer, or distributor or a nonprescription drug or commercially packed legend drug
or device;
(16) “legend drug” means a prescription drug;
(17) “manufacturing” means the production, preparation, propagation, conversion, or processing of a drug or
device, either directly or indirectly, by extraction from a substance of natural origin or independently by means of
chemical or biological synthesis, and includes packaging or repackaging of a substance or labeling or relabeling of
its container, and the promotion and marketing of drugs or devices; “manufacturing” also includes the preparation
and promotion of commercially available products from bulk compounds for resale by pharmacies, practitioners, or
other persons;
(18) “nonprescription drug” means a nonnarcotic medicine or drug that may be sold without a prescription and
that is prepackaged for use by the consumer and labeled in accordance with the requirements of the statutes and
regulations of the state and the federal government;
(19) “outpatient dispensing” means dispensing drugs for administration outside of the hospital pharmacy’s
control;
(20) “outsourcing facility” means a facility at one geographic location or address that is engaged in the
compounding of sterile drugs for a facility at another geographic location;
(21) “owner” means the owner of a place of business for wholesaling, retailing, compounding, or dispensing
drugs, medicines, or poisons;
(22) “patient counseling” means the communication by the pharmacist of information, as defined in the
regulations of the board, to the patient or care giver in order to improve therapy by ensuring proper use of drugs and
devices;
(23) “person” has the meaning given in AS 01.10.060 and also includes a governmental agency;
(24) “pharmaceutical care” is the provision of drug therapy and other pharmaceutical patient care services
intended to achieve outcomes related to the cure or prevention of a disease, elimination or reduction of a patient’s
symptoms, or arresting or slowing of a disease process as defined in regulations of the board;
(25) “pharmacist” means an individual currently licensed by this state to engage in the practice of pharmacy;
(26) “pharmacist-in-charge” means a pharmacist who accepts responsibility for operation of a pharmacy in a
manner that complies with laws and regulations applicable to the practice of pharmacy and the distribution of drugs
and who is personally in charge of the pharmacy and the pharmacy’s personnel;
(27) “pharmacy” means a place in this state where drugs are dispensed and pharmaceutical care is provided and
a place outside of this state that is subject to licensure or registration under AS 08.80.157(b);
(28) “pharmacy located outside of the state” means a pharmacy that prepares or mixes prescription drugs
outside of the state, regardless of the location at which those drugs may be shipped, mailed, or delivered to the
consumer;
(29) “pharmacy technician” means a supportive staff member who works under the immediate supervision of a
pharmacist;
(30) “practice of pharmacy” means the interpretation, evaluation, and dispensing of prescription drug orders in
the patient’s best interest; participation in drug and device selection, drug administration, drug regimen reviews, and
drug or drug-related research; provision of patient counseling and the provision of those acts or services necessary to
provide pharmaceutical care; the independent prescribing, dispensing, and administration of drugs in accordance
with AS 08.80.168; the responsibility for compounding and labeling of drugs and devices except labeling by a
manufacturer, repackager, or distributor of nonprescription drugs and commercially packaged legend drugs and
devices; proper and safe storage of drugs and devices; and maintenance of proper records for them;
(31) “practitioner” means an individual currently licensed, registered, or otherwise authorized by the
jurisdiction in which the individual practices to prescribe and administer drugs in the course of professional practice;
(32) “preceptor” means an individual who is currently licensed by the board, meets the qualifications as a
preceptor under the regulations of the board, and participates in the instructional training of pharmacy interns;
(33) “prescription drug” means a drug that, under federal law, before being dispensed or delivered, is required
to be labeled with either of the following statements: (A) “Caution: Federal law prohibits dispensing without
prescription”; (B) “Caution: Federal law restricts this drug to use by, or on the order of, a licensed veterinarian”; or a
drug that is required by an applicable federal or state law or regulation to be dispensed only under a prescription
drug order or is restricted to use by practitioners only;
(34) “prescription drug order” means a lawful order of a practitioner for a drug or device for a specific patient;
(35) “significant adverse drug reaction” means a drug-related incident that may result in serious harm, injury,
or death to the patient;
(36) “substitute” means to dispense, without the prescriber’s expressed authorization,
(A) an equivalent drug product in place of the prescribed drug; or
(B) an interchangeable biological product in place of the prescribed biological product;
(37) “third-party logistics provider” means an entity that provides or coordinates warehousing or other logistics
services for a product in interstate commerce on behalf of a manufacturer, wholesale distributor, or dispenser of the
product, and that does not take ownership of the product or have responsibility to direct the sale or disposition of the
product;
(38) “wholesale” means sale by a manufacturer, wholesale dealer, distributor, or jobber to a person who sells,
or intends to sell, directly to the user;
(39) “wholesale drug distributor” means anyone engaged in wholesale distribution of drugs, including
manufacturers; repackagers; own-label distributors; private label distributors; jobbers; brokers; warehouses,
including manufacturers’ and distributors’ warehouses; chain drug warehouses; wholesale drug warehouses;
independent wholesale drug traders; and retail pharmacies that conduct wholesale distributions.” “12 AAC 52.010. CLASSIFICATIONS OF LICENSURE.
Repealed 1/19/2024. ” “12 AAC 52.140. PHARMACY TECHNICIAN LICENSE.
(a) An applicant who meets the requirements on the checklist set out in (b) of this section has demonstrated the necessary qualifications for a pharmacy technician license. An applicant who does not meet the requirements on the checklist or whose responses on the form for application do not clearly show that the applicant is qualified to receive a pharmacy technician license will not be issued a license unless the board reviews the application and determines that the applicant meets the qualifications in this section for a pharmacy technician license.
(b) A pharmacy technician license will be issued to an applicant who
(1) submits a completed form for application, including
(A) the applicant’s name, mailing address, and telephone number; and
(B) the applicant’s date of birth that shows the applicant is at least 18 years of age, or at least 16 years of age and enrolled in a supervised high school, college, or apprenticeship pharmacy technician training program;
(2) certifies that the applicant has not been convicted of a felony or another crime that affects the applicant’s ability to perform the duties of a pharmacy technician safely and competently;
(3) certifies that the applicant has earned a high school diploma or its equivalent and provides the name of the issuing institution and the date the diploma or its equivalent was issued, or is enrolled in a supervised high school, college, or apprenticeship pharmacy technician training program;
(4) certifies that the applicant is fluent in the reading, writing, and speaking of the English language; and
(5) pays the applicable fees established in 12 AAC 02.310.
(c) Repealed 7/7/2022.
Authority: AS 08.80.005 AS 08.80.030” “12 AAC 52.230. PHARMACY TECHNICIANS.
(a) An individual who assists in performing functions
included in the definition of the practice of pharmacy must be licensed as a pharmacy technician.
(b) Before an individual may perform the tasks of a pharmacy technician, or functions in accordance with 12
AAC 52.235, the individual shall complete training required by the pharmacist-in-charge. Duties performed must be
consistent with the training received.
(c) Persons whose responsibilities are purely administrative, including bookkeepers, accountants, administrative
staff and persons who transport or deliver completed prescriptions to a patient or patient’s agent are not required to
obtain a pharmacy technician license.
Authority: AS 08.80.030 AS 08.80.480
12 AAC 52.235. PHARMACY TECHNICIAN WITH NATIONAL CERTIFICATION.
(a) A pharmacy technician who holds a national certification may, at the direction of the pharmacist on duty and under the direct supervision of that pharmacist,
(1) perform a final check of and distribute a non-controlled substance prescription if
(A) the prescription drug order has previously undergone a drug regimen review by a pharmacist, including determination of substitution;
(B) the pharmacy uses a bar code scanning and verification system that confirms that the drug selected to fill the prescription is the same as indicated on the prescription label;
(C) the pharmacy uses software that displays the image or graphical description of the correct drug being verified, or the institutional facility uses software that performs and verifies a barcode scan before administration; and
(D) each prescription distributed is electronically verified and the date and quantity distributed is documented in the patient record;
(2) transfer a non-controlled substance prescription drug order as described in 12 AAC 52.500;
(3) clarify or obtain missing information from the practitioner or the practitioner’s authorized agent on a noncontrolled substance prescription drug order; or
(4) administer a vaccine or related emergency medication in accordance with 12 AAC 52.992.
(b) Prescription drug order information clarifications under (a)(3) of this section must have the following information documented on the prescription drug order:
(1) the result of the clarification;
(2) the initials of the pharmacy technician who holds a national certification;
(3) the name of the practitioner or authorized agent that the pharmacy technician spoke to; and
(4) the date of the call.
(c) A pharmacy technician who holds a national certification may not sign or initial any document that is required to be signed or initialed by a pharmacist.
(d) In this section, “bar code scanning and verification system” means any technology that scans the bar code on a manufacturer drug container to ensure that the product being distributed matches the expectation of what was prescribed and inputted into the dispensing software.
Authority: AS 08.80.005 AS 08.80.030 AS 08.80.168” “12 AAC 52.300. LICENSE AND REGISTRATION RENEWAL.
(a) Pharmacy, remote pharmacy, wholesale drug distributor, outsourcing facility, third-party logistics provider, manufacturer, pharmacist, pharmacy technician,
and drug room licenses must be renewed biennially on or before a date set by the department.
(b) An applicant for renewal of a pharmacy, wholesale drug distributor, outsourcing facility, third-party logistics provider, manufacturer, or drug room license must submit on or before the license expiration date
(1) a completed renewal application on a form provided by the department;
(2) the license renewal fees required in 12 AAC 02.310; and
(3) an attestation that a self-inspection of the premises using the form provided by the department was completed within the last two years or since the last time the license was initially issued; the applicant must retain the self-inspection and make it available to the board upon request for the duration of the licensing period in which it was completed.
(c) An applicant for renewal of a pharmacist or pharmacy technician license shall submit on or before the license expiration date
(1) a completed renewal application on a form provided by the department;
(2) the license renewal fees required in 12 AAC 02.310; and
(3) an attestation that the applicant has met all continuing education requirements of 12 AAC 52.320 – 12 AAC 52.350.
(d) Repealed 7/1/2023.
(e) Repealed 7/1/2023.
(f) Repealed 7/1/2023.
(g) All renewal applications will be administratively processed and will not require board review unless the executive administrator has reason to believe that renewing the license poses an immediate threat to public health or safety.
Authority: AS 08.01.100 AS 08.80.030 AS 08.80.157
AS 08.80.005 AS 08.80.147 AS 08.80.165” “12 AAC 52.310. REINSTATEMENT OF A LAPSED PHARMACIST OR PHARMACY TECHNICIAN LICENSE.
(a) If a pharmacist’s or pharmacy technician’s license has lapsed for any reason, that pharmacist or pharmacy technician may not practice pharmacy until the board reinstates the license.
(b) The board will reinstate a pharmacist or pharmacy technician license that has been lapsed less than two years if the applicant submits
(1) a completed renewal application;
(2) any applicable license renewal fees required under 12 AAC 02.310; and
(3) documentation that the applicant has completed all continuing education requirements under 12 AAC
52.320 – 12 AAC 52.350 within the immediate two years before applying for reinstatement.
(c) The board will reinstate a pharmacist license that has been lapsed more than two years but less than five years if the applicant
(1) submits a completed application for reinstatement on a form provided by the department;
(2) pays any applicable license renewal fees required under 12 AAC 02.310 for the entire period the license
has been lapsed;
(3) submits documentation that the applicant has completed all continuing education requirements under 12 AAC 52.320 – 12 AAC 52.350 within the immediate two years before applying for reinstatement;
(4) qualifies by
(A) retaking and passing the examination required under 12 AAC 52.090(a); or
(B) providing verification that the applicant has continually practiced pharmacy in another state under a
license issued by the authority of that state for the period that the license has been lapsed; for purposes of AS 08.80.147 and this subparagraph, an applicant has continually practiced pharmacy if the pharmacist has actively practiced pharmacy in the other state for at least six months during each year that the license in the state was lapsed; and
(5) submits a verification issued directly to the board by each licensing jurisdiction where the applicant holds, or has ever held, a license as a pharmacist during the time period in which the applicant’s license was lapsed in the state that the applicant’s license in the other jurisdiction was not suspended, revoked, or otherwise restricted except for failure to apply for renewal or failure to obtain the required continuing education requirements.
(d) Repealed 8/1/2014.
(e) The board will not reinstate a pharmacy technician license that has been lapsed for two years or more.
(f) The board will not reinstate a pharmacist license that has been lapsed for five years or more.
Authority: AS 08.01.100 AS 08.80.030 AS 08.80.165
AS 08.80.005 AS 08.80.147” “12 AAC 52.325. CONTINUING EDUCATION REQUIREMENTS FOR PHARMACY TECHNICIANS.
(a) Except as provided in (c) of this section, an applicant for renewal of a pharmacy technician license shall certify that, during the concluding licensing period, the applicant
(1) completed 10 contact hours of continuing education accepted by the board under 12 AAC 52.340; or
(2) obtained initial certification as a pharmacy technician by the Pharmacy Technician Certification Board (PTCB).
(b) This section does not prevent the board from imposing additional continuing education requirements under its disciplinary powers.
(c) Instead of complying with the continuing education requirements in (a) of this section, an applicant for renewal of a pharmacy technician license for the first time may
(1) verify in an affidavit, on an application for renewal, that the applicant has read the state statutes and regulations compiled by the board; and
(2) submit an affidavit, signed by the pharmacist-in-charge, verifying the applicant’s pharmacy technician training in accordance with 12 AAC 52.230.
(d) An applicant for reinstatement of a pharmacy technician license that has expired shall certify that the applicant completed the continuing education requirements in (a) of this section before applying for reinstatement.
(e) A pharmacy technician who holds a national certification, before administering a vaccine or related emergency medication under 12 AAC 52.992, shall certify having completed one hour of ACPE-approved continuing education specific to immunizations or vaccines as part of the 10 contact hours of continuing education
required under (a) of this section.
Authority: AS 08.01.100 AS 08.80.030 AS 08.80.168
AS 08.80.005 AS 08.80.165” “12 AAC 52.340 APPROVED PROGRAMS.
(a) The following programs will be accepted by the board as continuing education for pharmacists and pharmacy technicians under 12 AAC 52.320 and 12 AAC 52.325:
(1) any program presented by a provider accredited by the ACPE that results in a continuing education certificate showing the date of the course and the ACPE Universal Activity Number associated with the program;
(2) cardiopulmonary resuscitation (CPR) courses presented by the American Red Cross or the American Heart Association that lead to CPR certification; the board will accept no more than one contact hour of continuing education credit in a 24 month period for completion of a CPR course.
(b) The following programs will be accepted by the board as continuing education under 12 AAC 52.325, when the subject contributes directly to the professional competency of a pharmacy technician and is directly related to pharmacy principles and practice:
(1) any program presented or approved by the Alaska Pharmacists Association;
(2) any program presented or approved by the Pharmacy Technician Certification Board (PTCB) or the National Pharmacy Technician Association (NPTA).
(c) An individual who presents an approved continuing education program may receive credit for the time spent during the actual presentation of the program. An individual may not receive credit for the same presentation more than once during a licensing period.
Authority: AS 08.80.005 AS 08.80.147 AS 08.80.165
AS 08.80.030
References
Statutes and Regulations December 2011
http://www.dced.state.ak.us/occ/pub/PharmacyStatutes.pdf
The data contained in this 2012 Annual Scorecard are accurate as of December 2012 . Because statutes and regulations are continually revised, the data are subject to change. These data have been verified with the state board of pharmacy. This scorecard is updated on an annual basis in order to incorporate statutory and regulatory changes. A new scorecard will be issued in July 2013.
Scoring rationale for Education and Training:
In order to protect the public and help ensure patient safety, it is important that pharmacy technicians are properly educated and trained. The most rigorous training is accredited training. The sole entity empowered to accredit pharmacy-technician training programs is the American Society of Health-System Pharmacists (ASHP). Please note that this is “programmatic accreditation” – not “institutional accreditation.” It is the content of the training program – as measured against established standards – that is being evaluated and accredited. Accredited training is vital to protecting patient safety because it means that a pharmacy-technician training program has met established quality standards to provide assurance and confidence to the public. For more information, please see http://www.ashp.org/menu/Accreditation/TechnicianAccreditation.aspx.
Scoring rationale for Certification:
Certification is the process by which a nongovernmental agency or association grants recognition to an individual who has met certain predetermined qualifications specified by that agency or association. This is often determined by an examination process. Numerous organizations have recommended that the certification exam conducted by the Pharmacy Technician Certification Board (PTCB) should be recognized as the sole, nationally-accredited certification exam for pharmacy technician certification – including the National Association of Boards of Pharmacy (NABP), the Texas State Board of Pharmacy (TSBP), and the U.S. Department of Veterans Affairs (VA). In a recent report, NABP recommended that states be encouraged to “recognize certification by the Pharmacy Technician Certification Board (PTCB).” Moreover, NABP performed a psychometric audit of the PTCB’s pharmacy technician certification examination (PTCE) in 2001 and determined that the PTCE is psychometrically sound, defensible, and valid. In May 2010, the TSBP awarded the PTCB with the Pharmacy Technician Certification Provider contract in Texas. PTCB was selected for the contract after a rigorous bidding and evaluation process that included formal reviews and evaluations from three independent psychometricians. TSBP confidently recognizes PTCB as the single provider of certification examinations for pharmacy technicians. In addition, in June 2010, the VA began requiring PTCB certification for VA pharmacy technicians employed at grade GS-6 and above.
Scoring rationale for Registration/Licensure:
Registration/licensure is the process by which the state maintains a list of all pharmacy technicians in the state and grants permission for an individual to work as a pharmacy technician in the state based on the applicant’s completion of all pre-requisites to registration/licensure – such as required training and certification.
Scoring rationale for Continuing Education:
Continuing education enables pharmacy technicians to fulfill their professional responsibility to maintain competence and up-to-date knowledge and skills in an environment of technological advances and increasingly complex, new medications and therapies.
Our Mission
The Emily Jerry Foundation is determined to help make our nation’s, world renowned, medical facilities safer for everyone, beginning with our babies and children. We are accomplishing this very important objective by focusing on increasing public awareness of key patient safety related issues and identifying technology and best practices that are proven to minimize the “human error” component of medicine. Through our ongoing efforts The Emily Jerry Foundation is working hard to save lives every day.
Recent Posts
Archives